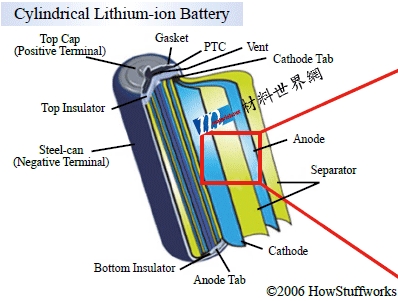
What is inside a lithium battery? |
|
Recently, many electric vehicle fires and explosions have once again pushed the safety of power batteries to the forefront. Peng Yumin, director of the Industrial Technology Research Institute, pointed out that the safety problems of lithium batteries mainly come from the increase in the internal temperature of the battery, including improper heating of the battery, overcharging, and short circuits caused by contact between the positive and negative materials, which will all cause the internal temperature of the lithium battery to rise. |
- Home
- FAQ
- FAQ1